Evaluation of hepatic lesions in livers from condemned pigs at the municipal slaughterhouse of Pasaje, Ecuador
Abstract
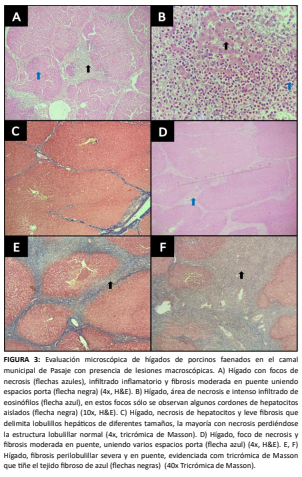
Ecuadorian pig farming is growing but faces significant sanitary challenges. Hepatic lesions detected in slaughtered pigs affect meat quality, food safety, and economic performance, highlighting management deficiencies and the need for specific local studies. The objective of this study was to characterize the macroscopic and microscopic lesions present in pig livers from animals slaughtered at the municipal abattoir of Pasaje, Ecuador. A total of 3,200 livers were inspected through official post-mortem evaluation, and 80 (2.5 %) showing visible alterations were selected for histopathological analysis. The main macroscopic lesions included hepatomegaly, discoloration, and fibrous adhesions, while microscopically, necrosis, inflammatory infiltration, fibrosis, hyperplasia, and congestion were frequently observed, showing patterns consistent with chronic liver damage. The marked presence of eosinophilic infiltrates associated with necrosis and bridging fibrosis suggests parasitic etiologies, likely linked to Ascaris suum infections and the development of “milk spot” lesions, recognized as a major cause of hepatic condemnation. These findings confirm that, although relatively low frequency, hepatic lesions represent a sensitive indicator for detecting health problems at the farm level and a source of economic loss, highlighting the role of post-mortem inspection as an epidemiological surveillance tool and the need to strengthen parasite control programs, biosecurity, and good management practices in pig production in the region.
Downloads
References
Acosta A, Cespedes-Cardenas N, Imbacuan C, Lentz HHK, Dietze K, Amaku M, Burbano A, Gonçalves VSP, Ferreira F. Modelling control strategies against classical swine fever: Influence of traders and markets using static and temporal networks in Ecuador. Prev. Vet. Med. [Internet]. 2022; 205:105683. doi: https://doi.org/qz9j DOI: https://doi.org/10.1016/j.prevetmed.2022.105683
Čobanović N, Janković L, Vasilev D, Dimitrijević M, Teodorović V, Kureljušić B, Karabasil N. Slaughterline records of various postmortem pathological lesions and their influence on carcass and meat quality in slaughtered pigs. Anim. Sci. J. [Internet]. 2019; 90(11):1475-1483. doi: https://doi.org/qz9k DOI: https://doi.org/10.1111/asj.13287
Jacobs P, Berends B, Lipman L. The value of current Ante Mortem meat inspection and food chain information of dairy cows in relation to Post Mortem findings and the protection of public health: A case for a more risk-based meat inspection. Foods. [Internet]. 2023; 12(3):616. doi: https://doi.org/qz9m DOI: https://doi.org/10.3390/foods12030616
Comin A, Jonasson A, Rockström U, Kautto AH, Keeling L, Nyman A-K, Lindberg A, Frössling J. Can we use meat inspection data for animal health and welfare surveillance? Front. Vet. Sci. [Internet]. 2023; 10:1129891. doi: https://doi.org/gstnbp DOI: https://doi.org/10.3389/fvets.2023.1129891
García-Díez J, Saraiva S, Moura D, Grispoldi L, Cenci-Goga BT, Saraiva C. The importance of the slaughterhouse in surveilling animals and public health: A systematic review. Vet. Sci. [Internet]. 2023; 10(2):167. doi: https://doi.org/qz9n DOI: https://doi.org/10.3390/vetsci10020167
Allievi C, Lana E, Rizzi R, Zanon A, Mortarino M, Manfredi MT. Abattoir monitoring of liver lesions caused by Ascaris suum larvae: A longitudinal study conducted in northern Italy. Prev. Vet. Med. [Internet]. 2025; 241:106553. doi: https://doi.org/qz9p DOI: https://doi.org/10.1016/j.prevetmed.2025.106553
Baquerre C, Montillet G, Pain B. Liver organoids in domestic animals: An expected promise for metabolic studies. Vet. Res. [Internet]. 2021; 52:47. doi: https://doi.org/grprdk DOI: https://doi.org/10.1186/s13567-021-00916-y
Valkova L, Voslarova E, Nincakova S, Passantino A, Vecerek V. The incidence of liver damage found during postmortem examination at the slaughterhouse. Animals. [Internet]. 2023; 13(5):839. doi: https://doi.org/qz9q DOI: https://doi.org/10.3390/ani13050839
Malhi H, Guicciardi ME, Gores GJ. Hepatocyte death: A clear and present danger. Physiol. Rev. [Internet]. 2010; 90(3):1165–1194. doi: https://doi.org/fqf3fh DOI: https://doi.org/10.1152/physrev.00061.2009
Olariu-Jurca A, Ilie MS, Lazău A, Ciulan V, Teslici LE, Avram E, Olariu-Jurca I. Morphopathological quantification of hepatopathies in necropsied swine. Agric. Agric. Sci. Procedia. [Internet]. 2015; 6:370–377. doi: https://doi.org/qz9r
Rosell C, Segalés J, Domingo M. Hepatitis and staging of hepatic damage in pigs naturally infected with porcine circovirus type 2. Vet. Pathol. [Internet]. 2000; 37(6):687–692. doi: https://doi.org/fmkwz5 DOI: https://doi.org/10.1354/vp.37-6-687
Vecerek V, Voslarova E, Semerad Z, Passantino A. The health and welfare of pigs from the perspective of post mortem findings in slaughterhouses. Animals. [Internet]. 2020; 10(5):825. doi: https://doi.org/g6xgmg DOI: https://doi.org/10.3390/ani10050825
Vitali M, Luppi A, Bonilauri P, Spinelli E, Santacroce E, Trevisi P. Benchmarking of anatomopathological lesions assessed at slaughter and their association with tail lesions and carcass traits in heavy pigs. Ital. J. Anim. Sci. [Internet]. 2021; 20(1):1103–1113. doi: https://doi.org/qz9s DOI: https://doi.org/10.1080/1828051X.2021.1944339
Ciui S, Morar A, Herman V, Tîrziu E, Imre M, Ban-Cucerzan A, Popa SA, Pătrînjan R-T, Morar D, Imre K. Causes of condemnations of edible parts of slaughtered pigs in Bavaria and their economic implications: A retrospective survey (2021–2022). Vet. Sci. [Internet]. 2025; 12(2):88. doi: https://doi.org/qz9t DOI: https://doi.org/10.3390/vetsci12020088
Olariu-Jurca A, Ilie MS, Lazău A, Ciulan V, Teslici LE, Avram E, Olariu-Jurca I. Morphopathological quantification of hepatopathies in necropsied swine. Agric. Agric. Sci. Procedia. [Internet]. 2015; 6:370–377. doi: https://doi.org/qz9r DOI: https://doi.org/10.1016/j.aaspro.2015.08.098
Zumbado L, de Oliveira JB, Chacón F, Hernández J, Quirós L, Murillo J. Identificación de parásitos gastrointestinales en granjas porcinas y pérdidas económicas por decomiso de hígados parasitados por Ascaris suum en mataderos de Costa Rica. Cienc. Vet. [Internet]. 2009; 27(1):7–21. Disponible en: https://goo.su/OEaLCe
Sánchez P, Pallarés FJ, Gómez MA, Bernabé A, Gómez S, Seva J. Importance of the knowledge of pathological processes for risk-based inspection in pig slaughterhouses (Study of 2002–2016). Asian-Australas. J. Anim. Sci. [Internet]. 2018; 31(11):1818–1827. doi: https://doi.org/gfh4wb DOI: https://doi.org/10.5713/ajas.18.0070
Liangpunsakul S, Chalasani N. Lipid mediators of liver injury in nonalcoholic fatty liver disease. Am. J. Physiol. Gastrointest. Liver Physiol. [Internet]. 2018; 316(1):G75–G81. doi: https://doi.org/gqjt6m DOI: https://doi.org/10.1152/ajpgi.00170.2018
Liang T, Alloosh M, Bell LN, Fullenkamp A, Saxena R, Van Alstine W, Bybee P, Werling K, Sturek M, Chalasani N, Masuoka HC. Liver injury and fibrosis induced by dietary challenge in the Ossabaw miniature swine. PLoS One. [Internet]. 2015; 10(5):e0124173. doi: https://doi.org/f7mnr5 DOI: https://doi.org/10.1371/journal.pone.0124173
Pedersen HD, Galsgaard ED, Christoffersen BØ, Cirera S, Holst D, Fredholm M, Latta M. NASH-inducing diets in Göttingen minipigs. J. Clin. Exp. Hepatol. [Internet]. 2020; 10(3):211–221. doi: https://doi.org/q2gb DOI: https://doi.org/10.1016/j.jceh.2019.09.004
Alizadeh A, Braber S, Akbari P, Garssen J, Fink-Gremmels J. Deoxynivalenol impairs weight gain and affects markers of gut health after low-dose, short-term exposure of growing pigs. Toxins. [Internet]. 2015; 7(6):2071-2095. doi: https://doi.org/f7pxv9 DOI: https://doi.org/10.3390/toxins7062071
Xue X, Wu P, Fan S, Yin Z, Zhang X. Proteomic analysis of liver injury induced by deoxynivalenol in piglets. Biology. [Internet]. 2025; 14(12):1721. doi: https://doi.org/q2gc DOI: https://doi.org/10.3390/biology14121721
Jeong JY, Kim J, Kim M, Park S. Efficacy of high-dose synbiotic additives for deoxynivalenol detoxification: Effects on blood biochemistry, histology, and intestinal microbiome in weaned piglets. Biology. [Internet]. 2024; 13(11):889. doi: https://doi.org/q2gd DOI: https://doi.org/10.3390/biology13110889
Mendes ÂJ, Ribeiro AI, Severo M, Niza-Ribeiro J. A multilevel study of the environmental determinants of swine ascariasis in England. Prev. Vet. Med. [Internet]. 2017; 148:10–20. doi: https://doi.org/q2gf DOI: https://doi.org/10.1016/j.prevetmed.2017.09.012
Fausto MC, Oliveira IC, Fausto GC, de Carvalho LM, Valente FL, Campos AK, de Araújo JV. Ascaris suum in pigs of the Zona da Mata, Minas Gerais State, Brazil. Rev. Bras. Parasitol. Vet. [Internet]. 2015; 24(3):375-378. doi: https://doi.org/q2gk DOI: https://doi.org/10.1590/S1984-29612015047
Wagner B, Polley L. Ascaris suum: Seasonal egg development rates in a Saskatchewan pig barn. Vet. Parasitol. [Internet]. 1999; 85(1):71–78. doi: https://doi.org/d7cgnj DOI: https://doi.org/10.1016/S0304-4017(99)00102-8
Knecht D, Popiołek M, Zaleśny G. Does meatiness of pigs depend on the level of gastro-intestinal parasites infection? Prev. Vet. Med. [Internet]. 2011; 99(2–4):234–239. doi: https://doi.org/bp4q6n DOI: https://doi.org/10.1016/j.prevetmed.2011.01.009
Knecht D, Jankowska A, Zaleśny G. The impact of gastrointestinal parasites infection on slaughter efficiency in pigs. Vet. Parasitol. [Internet]. 2012; 184(2-4):291–297. doi: https://doi.org/dr6jk9 DOI: https://doi.org/10.1016/j.vetpar.2011.09.006
Šoltésová H, Nagyová V, Tóthová C, Nagy O. Haematological and blood biochemical alterations associated with respiratory disease in calves. Acta Vet. Brno. [Internet]. 2015; 84(3):249–256. doi: https://doi.org/f7xm2q DOI: https://doi.org/10.2754/avb201584030249