A novel indirect ELISA for serological surveillance of fowl adenovirus in chickens: utilizing a regionally relevant field strain
Abstract
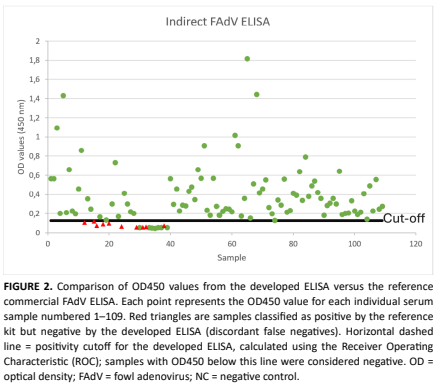
Fowl adenoviruses are significant avian pathogens that cause considerable economic losses globally, associated with diseases such as inclusion of body hepatitis, hydropericardium syndrome, and gizzard erosion. There is a critical need for rapid, sensitive, and high-throughput diagnostic tools for seroepidemiologic surveillance and evaluating vaccine efficacy, as the traditional diagnostic methods are limited due to the high viral diversity, which often hinders the effectiveness of group-specific targeted commercial ELISAs, as the antigens may not align with regionally prevalent serotypes. This study details the development and validation of an indirect ELISA designed for the detection of anti-Fowl adenoviruses antibodies in chickens, utilizing an antigen derived from a field strain of Fowl adenoviruses from Türkiye and propagated in cell culture. The assay was validated using 109 serum samples, benchmarked against the commercial BioCheck Fowl adenoviruses-1 ELISA CK132 kit. The developed ELISA demonstrated an optimal sample dilution of 1:400 and a cutoff value of 0.125. It achieved a sensitivity of 90.20 % and a specificity of 100 %. A Cohen’s kappa index of 0.675 indicated good agreement with the reference method. Furthermore, the assay exhibited good precision, with intra-assay coefficients of variation (CV %) ranging from 6.24 % to 11.03 % and an inter-assay CV % of 16.20 %, both within acceptable limits. This novel ELISA, employing a regionally relevant field strain, offers a cost-effective and specific tool for Fowl adenoviruses serological surveillance and vaccine monitoring, particularly beneficial in regions where circulating strains vary from vaccine strains.
Downloads
References
Fitzgerald SD, Rautenschlein S, Mahsoub HM, Pierson FW, Reed WM, Jack SW. Adenovirus infections. In: Swayne DE, Boulianne M, Logue CM, McDougald LR, Nair V, Suarez DL, Wit S, Grimes T, Johnson D, Kromm M, Prajitno TY, Rubinoff I, Zavala G, editors. Diseases of Poultry. 14th edition. Hoboken (NJ): John Wiley & Sons; 2020. p. 321-363. doi: https://doi.org/qwgv
Hess M. Detection and differentiation of avian adenoviruses: a review. Avian Pathol. [Internet]. 2000; 29(3):195-206. doi: https://doi.org/bbzhjs DOI: https://doi.org/10.1080/03079450050045440
Harrach B, Benkő M. Adenoviruses (Adenoviridae). In: Bamford DH, Zuckerman M, editors. Encyclopedia of Virology (4th ed). Oxford (UK): Academic Press; 2021 [cited 20 Aug 2025]. p. 3-16. Available in: https://goo.su/dplSNtO DOI: https://doi.org/10.1016/B978-0-12-814515-9.00057-6
Sohaimi NM, Hair-Bejo M. A recent perspective on fiber and hexon genes proteins analyses of fowl adenovirus toward virus infectivity—A review. Open Vet. J. 2021; 11(4):569-580. doi: https://doi.org/g8p8fj DOI: https://doi.org/10.5455/OVJ.2021.v11.i4.6
Huang J, Tan D, Wang Y, Liu C, Xu J, Wang J. Egg drop syndrome virus enters duck embryonic fibroblast cells via clathrin-mediated endocytosis. Virus Res. [Internet]. 2015; 210:69-76. doi: https://doi.org/f7z26k DOI: https://doi.org/10.1016/j.virusres.2015.07.014
Matczuk AK, Niczyporuk JS, Kuczkowski M, Woźniakowski G, Nowak M, Wieliczko A. Whole genome sequencing of Fowl aviadenovirus A - a causative agent of gizzard erosion and ulceration, in adult laying hens. Infec. Genet. Evol. [Internet]. 2017; 48:47-53. doi: https://doi.org/f9x4pz DOI: https://doi.org/10.1016/j.meegid.2016.12.008
Morshed R, Hosseini H, Langeroudi AG, Fard MHB, Charkhkar S. Fowl Adenoviruses D and E cause inclusion body hepatitis outbreaks in broiler and broiler breeder pullet flocks. Avian Dis. [Internet]. 2017; 61(2):205-210. doi: https://doi.org/g8hcsp DOI: https://doi.org/10.1637/11551-120516-Reg.1
Pan Q, Wang J, Gao Y, Cui H, Liu C, Qi X, Zhang Y, Wang Y, Li K, Gao L, Wang X. Development and application of a novel ELISA for detecting antibodies against group I fowl adenoviruses. Appl. Microbiol. Biotechnol. [Internet]. 2020; 104:853-859. doi: https://doi.org/qwgw DOI: https://doi.org/10.1007/s00253-019-10208-3
Feichtner F, Schachner A, Berger E, Hess M. Fiber-based fluorescent microsphere immunoassay (FMIA) as a novel multiplex serodiagnostic tool for simultaneous detection and differentiation of all clinically relevant fowl adenovirus (FAdV) serotypes. J. Immunol. Methods. [Internet]. 2018; 458:33-43. doi: https://doi.org/gdqrz2 DOI: https://doi.org/10.1016/j.jim.2018.03.002
Şahindokuyucu İ, Çöven F, Kılıç H, Yılmaz Ö, Kars M, Yazıcıoğlu Ö, Ertunç E, Yazıcı Z. First report of fowl aviadenovirus serotypes FAdV-8b and FAdV-11 associated with inclusion body hepatitis in commercial broiler and broiler-breeder flocks in Turkey. Arch. Virol. [Internet]. 2020; 165:43-51. doi: https://doi.org/qwgx DOI: https://doi.org/10.1007/s00705-019-04449-w
Cizmecigil UY, Umar S, Yilmaz A, Bayraktar E, Turan N, Tali B, Aydin O, Tali HE, Yaramanoglu M, Yilmaz SG, Kolukisa A, Sadeyen JR, Iqbal M, Yilmaz H. Characterization of fowl adenovirus (FAdV-8b) strain concerning the geographic analysis and pathological lesions associated with inclusion body hepatitis in broiler flocks in Turkey. J. Vet. Res. [Internet]. 2020; 64(2):231-237. doi: https://doi.org/qwgz DOI: https://doi.org/10.2478/jvetres-2020-0026
Günes A, Marek A, Grafl B, Berger E, Hess M. Real-time PCR assay for universal detection and quantitation of all five species of fowl adenoviruses (FAdV-A to FAdV-E). J. Virol. Methods. [Internet]. 2012; 183(2):147-153. doi: https://doi.org/f32kw4 DOI: https://doi.org/10.1016/j.jviromet.2012.04.005
Pan Q, Wang J, Gao Y, Cui H, Liu C, Qi X, Zhang Y, Wang Y, Wang X. Identification of two novel fowl adenovirus C-specific B cell epitopes using monoclonal antibodies against the capsid hexon protein. Appl. Microbiol. Biotechnol. [Internet]. 2018; 102(21):9243-9253. doi: https://doi.org/gfmjz3 DOI: https://doi.org/10.1007/s00253-018-9262-4
Yuan XY, Wang YL, Meng K, Zhang YX, Xu HY, Ai W. LAMP real-time turbidity detection for fowl adenovirus. BMC Vet. Res. [Internet]. 2019; 15(1):256. doi: https://doi.org/qwg2 DOI: https://doi.org/10.1186/s12917-019-2015-5
Li PH, Zheng P, Zhang TF, Wen GP, Shao H, Luo Q. Fowl adenovirus serotype 4: Epidemiology, pathogenesis, diagnostic detection, and vaccine strategies. Poultry Sci. [Internet]. 2017; 96(8):2630-2640. doi: https://doi.org/gbrjgn DOI: https://doi.org/10.3382/ps/pex087
Rajasekhar R, Roy P. Recombinant hexon antigen based single serum dilution ELISA for rapid serological profiling against fowl adenovirus-4 causing hydropericardium syndrome in chickens. J. Virol. Methods. [Internet]. 2014; 207:121-127. doi: https://doi.org/f6h8vx DOI: https://doi.org/10.1016/j.jviromet.2014.06.017
Ather F, Zia MA, Habib M, Shah MS. Development of an ELISA for the detection of fowl adenovirus serotype-4 utilising fiber protein. Biologicals. [Internet]. 2024; 85:101752. doi: https://doi.org/qwg3 DOI: https://doi.org/10.1016/j.biologicals.2024.101752
Lim SY, Bauermeister A, Kjonaas RA, Ghosh SK. Phytol-based novel adjuvants in vaccine formulation: 2. Assessment of efficacy in the induction of protective immune responses to lethal bacterial infections in mice. J. Immune Based Therap. Vaccines. [Internet]. 2006; 4:5. doi: https://doi.org/dtcjsv DOI: https://doi.org/10.1186/1476-8518-4-5
Kumar S, Chaturvedi V, Kumar B, Kumar P, Somarajan S, Kumar A, Yadav A, Sharma B. Improved humoral immune response of oil adjuvant vaccine by saponin coadjuvantation against haemorrhagic septicaemia in mice and buffalo calves. Indian J. Anim. Sci. [Internet]. 2012; 82(9):953-957. doi: https://doi.org/qwg4 DOI: https://doi.org/10.56093/ijans.v82i9.23644
Sayin Z, Erganiş O. Diagnosis of bovine tuberculosis by PPD-ELISA and sonication-ELISA. Isr. J. Vet. Med. [Internet]. 2013 [cited 23 Sept 2025]; 68(3):180-184. Available in: https://goo.su/OCxhI
Erganiş O, Hadimli HH, Kav K, Sakmanoğlu A, Sayın Z, Pınarkara Y. Farelerde ve koyunlarda inaktif Corynebacterium pseudotuberculosis aşısının kazeöz lenfadenitise karşı etkinliği. Eurasian J Vet Sci. [Internet]. 2014 [cited 22 Sept 2025]; 30(2):72-79. Available in: https://goo.su/XQfT48t
Ferrero I, Dewilde S, Poletti P, Canepa B, Giachino E, Dall’Ara P, Filipe J. Development of a new indirect ELISA test for the detection of anti-Feline Coronavirus antibodies in cats. Vet. Sci. [Internet]. 2025; 12(3):245. doi: https://doi.org/qwg5 DOI: https://doi.org/10.3390/vetsci12030245
Yuan F, Chen C, Covaleda LM, Martins M, Reinhart JM, Sullivan DR, Diel DG, Fang Y. Development of monoclonal antibody-based blocking ELISA for detecting SARS-CoV-2 exposure in animals. mSphere. [Internet]. 2023; 8(4):e00067-23. doi: https://doi.org/qwg6 DOI: https://doi.org/10.1128/msphere.00067-23
Zheng Z, Houchins D, Ung J, Richard JL. Validation of an ELISA test kit for the detection of deoxynivalenol in several food commodities. JSM Mycotoxins. [Internet]. 2003; 2003(Suppl 3):295-302. doi: https://doi.org/qwg7 DOI: https://doi.org/10.2520/myco1975.2003.Suppl3_295
Mei L, Wu P, Ye J, Gao G, Shao L, Huang S, Li Y, Yang X, Chen H, Cao S. Development and application of an antigen capture ELISA assay for diagnosis of Japanese encephalitis virus in swine, human and mosquito. Virol. J. [Internet]. 2012; 9:4. doi: https://doi.org/fx74kk DOI: https://doi.org/10.1186/1743-422X-9-4
Appeltrath GA, Parreuter J, Lindemann M, Klump H, Karsten CB. An efficient ELISA protocol for measurement of SARS-CoV-2 spike-specific IgG in human plasma and serum samples. MethodsX. [Internet]. 2024; 12:102596. doi: https://doi.org/qwg8 DOI: https://doi.org/10.1016/j.mex.2024.102596
Kovařčík K. The development and application of an indirect ELISA test for the detection of antibodies to bovine respiratory syncytial virus in blood serum. Vet. Med. Czech. [Internet]. 2001; 46(2):29-34. doi: https://doi.org/qwg9 DOI: https://doi.org/10.17221/7848-VETMED
Villanueva-Saz S, Basurco A, Martín V, Fernández A, Loste A, Verde MT. Comparison of a qualitative immunochromatographic test with two quantitative serological assays for the detection of antibodies to Leishmania infantum in dogs. Acta Vet. Scand. [Internet]. 2019; 61:38. doi: https://doi.org/qwhb DOI: https://doi.org/10.1186/s13028-019-0473-1
Shao H, Wang P, Wang W, Zhang J, Li T, Liang G, Gao W, Qin A, Ye J. A novel monoclonal antibodies-based sandwich ELISA for detection of serotype 4 fowl adenovirus. Avian Pathol. [Internet]. 2019; 48(3):204-208. doi: https://doi.org/qwhc DOI: https://doi.org/10.1080/03079457.2019.1566595
Lee JW, Devanarayan V, Barrett YC, Weiner R, Allinson J, Fountain S, Keller S, Weinryb I, Green M, Duan L, Rogers JA, Millham R, O’Brien PJ, Sailstad JM, Khan MN, Ray C, Wagner JA. Fit-for-purpose method development and validation for successful biomarker measurement. Pharm. Res. [Internet]. 2006; 23(2):312-328. doi: https://doi.org/fp296j DOI: https://doi.org/10.1007/s11095-005-9045-3
Kelley M, DeSilva B. Key elements of bioanalytical method validation for macromolecules. AAPS J. [Internet]. 2007; 9(2):17. doi: https://doi.org/cm9wqt DOI: https://doi.org/10.1208/aapsj0902017
Lexmond WS, Mee JVD, Ruiter F, Platzer B, Stary G, Yen EH, Dehlink E, Nurko S, Fiebiger E. Development and validation of a standardised ELISA for the detection of soluble Fc-epsilon-RI in human serum. J. Immunol. Methods. [Internet]. 2011; 373(1-2):192-199. doi: https://doi.org/cpt3kc DOI: https://doi.org/10.1016/j.jim.2011.08.018
Varotto-Boccazzi I, Manenti A, Dapporto F, Gourlay LJ, Bisaglia B, Gabrieli P, Forneris F, Faravelli S, Bollati V, Rubolini D, Zuccotti G, Montomoli E, Epis S, Bandi C. Epidemic preparedness—Leishmania tarentolae as an easy-to-handle tool to produce antigens for viral diagnosis: application to COVID-19. Front. Microbiol. [Internet]. 2021; 12:736530. doi: https://doi.org/qwhd DOI: https://doi.org/10.3389/fmicb.2021.736530
Valero MA, Periago MV, Pérez-Crespo I, Rodríguez E, Perteguer MJ, Gárate T, González-Barberá EM, Mas-Coma S. Assessing the validity of an ELISA test for the serological diagnosis of human fascioliasis in different epidemiological situations. Trop. Med. Int. Health. [Internet]. 2012; 17(5):630-636. doi: https://doi.org/dm8c DOI: https://doi.org/10.1111/j.1365-3156.2012.02964.x
Lu H, Wang W, Zhang J, Shao H, Li L, Li T, Xie Q, Wan Z, Qin A, Ye J. An efficient fiber-based ELISA for detection of antibody against fowl adenovirus serotypes 7 and 8. J. Vet. Diagn. Invest. [Internet]. 2020; 32(3):444-449. doi: https://doi.org/qwhf DOI: https://doi.org/10.1177/1040638720913354
Junnu S, Lertwatcharasarakul P, Jala S, Phattanakunanan S, Moonjit P, Songserm T. Developing an indirect ELISA based on recombinant hexon protein for serological detection of inclusion body hepatitis in chickens. J. Vet. Med. Sci. [Internet]. 2014; 76(2):289-293. doi: https://doi.org/f5x3v2 DOI: https://doi.org/10.1292/jvms.13-0196
Hosamani M, Basagoudanavar SH, Tamil-Selvan RP, Das V, Ngangom P, Sreenivasa BP, Hegde R, Venkataramanan R. A multi-species indirect ELISA for detection of non-structural protein 3ABC specific antibodies to foot-and-mouth disease virus. Arch. Virol. [Internet]. 2015; 160(4):937–944. doi: https://doi.org/f65jjp DOI: https://doi.org/10.1007/s00705-015-2339-9
Calnek BW, Shek WR, Menendez NA, Stiube P. Serological cross-reactivity of Avian Adenovirus serotypes in an enzyme-linked immunosorbent assay. Avian. Dis. [Internet]. 1982; 26(4):897-906. doi: https://doi.org/b22rtt DOI: https://doi.org/10.2307/1589878