Efectos terapéuticos del salidrosido en el síndrome de ascitis de pollos de engorde a través de la vía PI3K/AKT/mTOR
Resumen
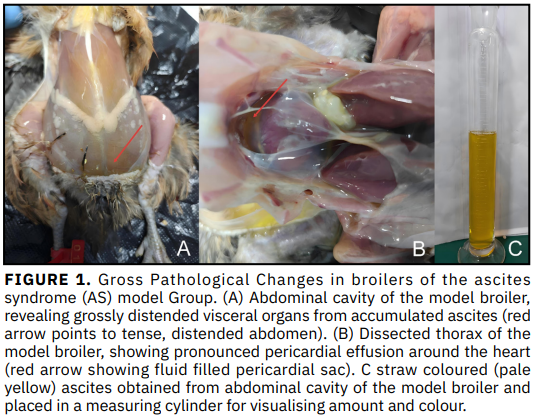
El presente estudio tuvo como objetivo examinar el impacto del salidrósido en la expresión de PI3K, AKT y mTOR en el tejido pulmonar de pollos de engorde afectados por el síndrome ascítico, así como dilucidar su posible mecanismo de acción contra esta patología. Noventa pollos Qingjiao Ma fueron asignados aleatoriamente a tres grupos de treinta individuos cada uno: grupo modelo, grupo de tratamiento y grupo control. Para inducir el síndrome ascítico, tanto el grupo modelo como el de tratamiento recibieron una dieta basal enriquecida con alto contenido de grasa (5 % de aceite de colza), alta proteína (4 % de harina de pescado) y alto sodio (0,12 % de NaCl en el agua de bebida) desde el día 1 de edad hasta los 42 días de edad. El grupo de tratamiento recibió adicionalmente un 0,2 % de salidrósido en su dieta. Todos los pollos fueron sacrificados a los 42 días de edad, y se recolectó tejido pulmonar para los análisis posteriores. Los niveles de expresión de ARNm y proteínas de PI3K, AKT y mTOR en los tejidos pulmonares se analizaron mediante técnicas de qRT-PCR, IHC y WB; para el análisis estadístico se aplicó el análisis de varianza de una vía (one-way ANOVA), estableciendo el nivel de significancia en P < 0,05. En comparación con el grupo control, la expresión de PI3K, AKT y mTOR —a nivel de ARNm y proteína, incluidas sus formas fosforiladas— se encontró muy regulada al alza en los pollos de engorde con síndrome ascítico inducido (P < 0,05). El tratamiento con salidrósido revertirá estos cambios al reducir las expresiones génica y proteica de dichas moléculas señalizadoras en relación con el grupo modelo (P< 0,05). La vía de transducción de señales PI3K/AKT/mTOR participa en la aparición y progresión del síndrome ascítico, y el salidrósido probablemente ejerce efectos terapéuticos sobre esta enfermedad mediante la inhibición de dicha vía de señalización.
Descargas
Citas
Cheng S, Liu X, Liu P, Li G, Guo X, Hu G, Li L, Wu C, Xu Z, Zhou Q, Jiang J, Luo S, Huang H, Ping L. Dysregulated expression of mRNA and SNP in pulmonary artery remodeling in ascites syndrome in broilers. Poult. Sci. [Internet]. 2021; 100(3):100877. URL: https://doi.org/gp4s3t DOI: https://doi.org/10.1016/j.psj.2020.11.054
Chen J, Jiang C, Hu X, Zhang Y, Gao X, Guo X, Jin H, Zhang Y, Wu Y, Liang J, Liu P. Mechanism of pulmonary arterial vascular cell dysfunction in pulmonary hypertension in broiler chickens. Avian Pathol. [Internet]. 2025; 54(5):548-559. URL: https://doi.org/qw58 DOI: https://doi.org/10.1080/03079457.2025.2480802
Rahimi M, Rahimi S, Karimi-Torshizi MA, Sharafi M, Masoudi AA, Grimes JL. Peroxisome proliferator-activated receptor gamma (PPARγ) activation: a potential treatment for ascites syndrome in broiler chickens. Poult. Sci. [Internet]. 2023; 102(9):102859. doi: https://doi.org/qw59 DOI: https://doi.org/10.1016/j.psj.2023.102859
Fang W, Wang E, Liu P, Gao X, Hou X, Hu G, Li G, Cheng J, Jiang C, Yan L, Wu C, Xu Z, Liu P. The relativity analysis of hypoxia inducible factor-1a in pulmonary arterial hypertension (ascites syndrome) in broilers: a review. Avian Pathol. [Internet]. 2024; 53(6):441-450. URL: https://doi.org/qw6b DOI: https://doi.org/10.1080/03079457.2024.2358882
Yu J, Liu X, Wang K, Wang H, Han Y, Kang J, Deng R, Zhou H, Duan Z. Underlying mechanism of Qiling Jiaogulan Powder in the treatment of broiler ascites syndrome. Poult. Sci. [Internet]. 2023; 102(1):102144. URL: https://doi.org/g62mdh DOI: https://doi.org/10.1016/j.psj.2022.102144
Pesti-Asboth G, Szilagyi E, Birone-Molnar P, Olah J, Babinszky L, Czegledi L, Cziaky Z, Paholcsek M, Stundl L, Remenyik J. Monitoring physiological processes of fast-growing broilers during the whole life cycle: Changes of redox-homeostasis effected to trassulfuration pathway predicting the development of non-alcoholic fatty liver disease. PLoS One. [Internet]. 2023; 18(8):290310. URL: https://doi.org/qw6c DOI: https://doi.org/10.1371/journal.pone.0290310
Pena A, Kobir A, Goncharov D, Goda A, Kudryashova TV, Ray A, Vanderpool R, Baust J, Chang B, Mora AL, Gorcsan JR, Goncharova EA. Pharmacological Inhibition of mTOR Kinase Reverses Right Ventricle Remodeling and Improves Right Ventricle Structure and Function in Rats. Am. J. Respir. Cell Mol. Biol. [Internet]. 2017; 57(5):615-625. URL: https://doi.org/gch2fv DOI: https://doi.org/10.1165/rcmb.2016-0364OC
Li Y, Li C, Xu W, Zhao J, Liu K, Liu X, Li Y, Tang Z, Li A, Zhang H. Chondroitin sulfate reverses tibial dyschondroplasia, broiler chondrocyte proliferation and differentiation dysfunction via the CHST11/ẞ-Catenin pathway. Int. J. Biol. Macromol. [Internet]. 2025; 315(2):144488. URL: https://doi.org/qw6g DOI: https://doi.org/10.1016/j.ijbiomac.2025.144488
Tarrant KJ, Dey S, Kinney R, Anthony NB, Rhoads DD. Multi-generational genome wide association studies identify chromosomal regions associated with ascites phenotype. Poult. Sci. [Internet]. 2017; 96(6):1544-1552. URL: https://doi.org/gbhhwf DOI: https://doi.org/10.3382/ps/pew478
Li Y, Yi J, Liu K, Liu X, Yangzom C, Pan J, Iqbal M, Hu L, Tang Z, Li Y, Zhang H. Mn2O3 NPs-induced liver injury is potentially associated with gut microbiota dysbiosis in broiler chicken. Food Chem. Toxicol. [Internet]. 2025; 202:115487. URL: https://doi.org/qw6h DOI: https://doi.org/10.1016/j.fct.2025.115487
Neong SF, Adebayo D, Wong F. An update on the pathogenesis and clinical management of cirrhosis with refractory ascites. Expert Rev. Gastroenterol. Hepatol. [Internet]. 2019; 13(4):293-305. URL: https://doi.org/qw6k DOI: https://doi.org/10.1080/17474124.2018.1555469
Hou J, Wu P, Cai J, Xia B, Lei Y, Huang C, Li Y, Tareen MI, Tang Z, Zhang H. Gut microbiota dysbiosis amplifies thiram hepatotoxicity via a mitochondrial-autophagy-apoptosis nexus orchestrated by the gut-liver axis. Cell. Signal. [Internet]. 2025; 136:112104. URL: https://doi.org/qw6m DOI: https://doi.org/10.1016/j.cellsig.2025.112104
Khodambashi-Emami N, Golian A, Danesh-Mesgaran M, Anthony NB, Rhoads DD. Mitochondrial biogenesis and PGC-1alpha gene expression in male broilers from ascites-susceptible and -resistant lines. J. Anim. Physiol. Anim. Nutr. [Internet]. 2018; 102 (1):482-485. URL: https://doi.org/gdkdm9 DOI: https://doi.org/10.1111/jpn.12706
Alzahrani AS. PI3K/Akt/mTOR inhibitors in cancer: At the bench and bedside. Semin. Cancer Biol. [Internet]. 2019; 59:125-132. URL: https://doi.org/gknpzr DOI: https://doi.org/10.1016/j.semcancer.2019.07.009
Wu S, Liu K, Huang X, Sun Q, Wu X, Mehmood K, Li Y, Zhang H. Molecular mechanism of miR-203a targeting Runx2 to regulate thiram induced-chondrocyte development. Pestic. Biochem. Physiol. [Internet]. 2024; 200:105817. URL: https://doi.org/qw6n DOI: https://doi.org/10.1016/j.pestbp.2024.105817
Wang X, Jian W, Luo Q, Fang L. CircSEMA4B inhibits the progression of breast cancer by encoding a novel protein SEMA4B-211aa and regulating AKT phosphorylation. Cell Death Dis. [Internet]. 2022; 13(9):794. URL: https://doi.org/qw6p DOI: https://doi.org/10.1038/s41419-022-05246-1
Zhou W, Li W, Wang S, Salovska B, Hu Z, Tao B, Di Y, Punyamurtula U, Turk BE, Sessa WC, Liu Y. An optogenetic-phosphoproteomic study reveals dynamic Akt1 signaling profiles in endothelial cells. Nat. Commun. [Internet]. 2023; 14(1):3803. URL: https://doi.org/qw6q DOI: https://doi.org/10.1038/s41467-023-39514-1
Huang X, Liu X, Xu W, Li Y, Wu P, He X, Zhong D, Ataya FS, Li Y. Expression of Pyruvate Dehydrogenase Kinase (PDK) in Lungs during Progression of Pulmonary Hypertension Syndrome in Broilers. Pak. Vet. J. [Internet]. 2024; 44(2):286-291. URL: https://doi.org/qw6r
Tang H, Gupta A, Morrisroe SA, Bao C, Schwantes-An T, Gupta G, Liang S, Sun Y, Chu A, Luo A, Ramamoorthi-Elangovan V, Sangam S, Shi Y, Naidu SR, Jheng J, Ciftci-Yilmaz S, Warfel NA, Hecker L, Mitra S, Coleman AW, Lutz KA, Pauciulo MW, Lai Y, Javaheri A, Dharmakumar R, Wu W, Flaherty DP, Karnes JH, Breuils-Bonnet S, Boucherat O, Bonnet S, Yuan JX, Jacobson JR, Duarte JD, Nichols WC, Garcia JGN, Desai AA. Deficiency of the Deubiquitinase UCHL1 Attenuates Pulmonary Arterial Hypertension. Circulation. [Internet]. 2024; 150(4):302-316. URL: https://doi.org/gt4gsc DOI: https://doi.org/10.1161/CIRCULATIONAHA.123.065304
Sun Q, Wu S, Liu K, Li Y, Mehmood K, Nazar M, Hu L, Pan J, Tang Z, Liao J, Zhang H. miR-181b-1-3p affects the proliferation and differentiation of chondrocytes in TD broilers through the WIF1/Wnt/ẞ-catenin pathway. Pestic. Biochem. Physiol. [Internet]. 2023; 197:105649. URL: https://doi.org/qw6s DOI: https://doi.org/10.1016/j.pestbp.2023.105649
Gui D, Cui Z, Zhang L, Yu C, Yao D, Xu M, Chen M, Wu P, Li G, Wang L, Huang X. Salidroside attenuates hypoxia-induced pulmonary arterial smooth muscle cell proliferation and apoptosis resistance by upregulating autophagy through the AMPK-mTOR-ULK1 pathway. BMC Pulm. Med. [Internet]. 2017; 17(1):191. URL: https://doi.org/gsh6j4 DOI: https://doi.org/10.1186/s12890-017-0477-4
Liu R, Ma T, Yang Q, Xiao W, Yin L, Yin M, Zhang J, Wang C. Salidroside suppresses proliferation and migration in prostate cancer via the PI3K/AKT pathway. Cancer Biomark. [Internet]. 2023; 38(3):321-332. URL: https://doi.org/gt47xr DOI: https://doi.org/10.3233/CBM-220454
Gan W, Dai X, Dai X, Xie J, Yin S, Zhu J, Wang C, Liu Y, Guo J, Wang M, Liu J, Hu J, Quinton RJ, Ganem NJ, Liu P, Asara JM, Pandolfi PP, Yang Y, He Z, Gao G, Wei W. LATS suppresses mTORC1 activity to directly coordinate Hippo and mTORC1 pathways in growth control. Nat. Cell Biol. [Internet]. 2020; 22(2):246-256. doi: https://doi.org/gq74x2 DOI: https://doi.org/10.1038/s41556-020-0463-6
Li Y, Yang L, Dong L, Yang Z, Zhang J, Zhang S, Niu M, Xia J, Gong Y, Zhu N, Zhang X, Zhang Y, Wei X, Zhang Y, Zhang P, Li S. Crosstalk between the Akt/mTORC1 and NF-KB signaling pathways promotes hypoxia-induced pulmonary hypertension by increasing DPP4 expression in PASMCs. Acta Pharmacol. Sin. [Internet]. 2019; 40(10):1322-1333. URL: https://doi.org/qw6t DOI: https://doi.org/10.1038/s41401-019-0272-2
Li S, Ji Y, Zhu S, Liu M, Luo D, Luo Q, Mo M, Long H, Peng F, Jia Z, Dou X. Integrating single-cell RNA-seq, bulk RNA-seq and network pharmacology reveals protective effect of salidroside in peritoneal dialysis-associated peritoneal fibrosis. Front. Pharmacol. [Internet]. 2025; 16:1558366. URL: https://doi.org/qw6v DOI: https://doi.org/10.3389/fphar.2025.1558366
Teng L, Gao J, Zhou L, Xian Q, Li J, Yang SJ. Influence of salidroside on expression level of endothelin-1 and its receptors under hypoxic conditions in chicken embryonic pulmonary artery smooth muscle cells. Pak. Vet. J. [Internet]. 2016 [cited 22 Sept 2025]; 36:214-218. URL: https://goo.su/VfZjnWZ
Dai Z, Wang H, Liu J, Zhang H, Li Q, Yu X, Zhang R, Yang C. Comparison of the Effects of Yucca saponin, Yucca schidigera, and Quillaja saponaria on Growth Performance, Immunity, Antioxidant Capability, and Intestinal Flora in broilers. Animals. [Internet]. 2023; 13(9):1447. URL: https://doi.org/qw6w DOI: https://doi.org/10.3390/ani13091447
Li X, Li J, Yuan H, Chen Y, Li S, Jiang S, Zha Xi Y, Zhang G, Lu J. Effect of supplementation with Glycyrrhiza uralensis extract and Lactobacillus acidophilus on growth performance and intestinal health in broiler chickens. Front. Vet. Sci. [Internet]. 2024; 11:1436807. URL: https://doi.org/qw6x DOI: https://doi.org/10.3389/fvets.2024.1436807
Zhang Y, Ge H, Yu Y, Gao H, Fan X, Li Q, Zhou Z. Dietary salidroside supplementation improves meat quality and antioxidant capacity and regulates lipid metabolism in broilers. Food Chem. X. [Internet]. 2024; 22:101406. URL: https://doi.org/qw6z DOI: https://doi.org/10.1016/j.fochx.2024.101406
Yanagihara T, Tsubouchi K, Zhou Q, Chong M, Otsubo K, Isshiki T, Schupp JC, Sato S, Scallan C, Upagupta C, Revill S, Ayoub A, Chong SG, Dvorkin-Gheva A, Kaminski N, Tikkanen J, Keshavjee S, Paré G, Guignabert C, Ask K, Kolb MRJ. Vascular-Parenchymal Cross-Talk Promotes Lung Fibrosis through BMPR2 Signaling. Am. J. Respir. Crit. Care Med. [Internet]. 2023; 207(11):1498-1514. doi: https://doi.org/gr5c9z DOI: https://doi.org/10.1164/rccm.202109-2174OC
Xue R, Wang J, Yang L, Liu X, Gao Y, Pang Y, Wang Y, Hao J. Coenzyme Q10 Ameliorates Pancreatic Fibrosis via the ROS-Triggered mTOR Signaling Pathway. Oxid. Med. Cell. Longev. [Internet]. 2019; 2019:8039694. URL: https://doi.org/qw64 DOI: https://doi.org/10.1155/2019/8039694